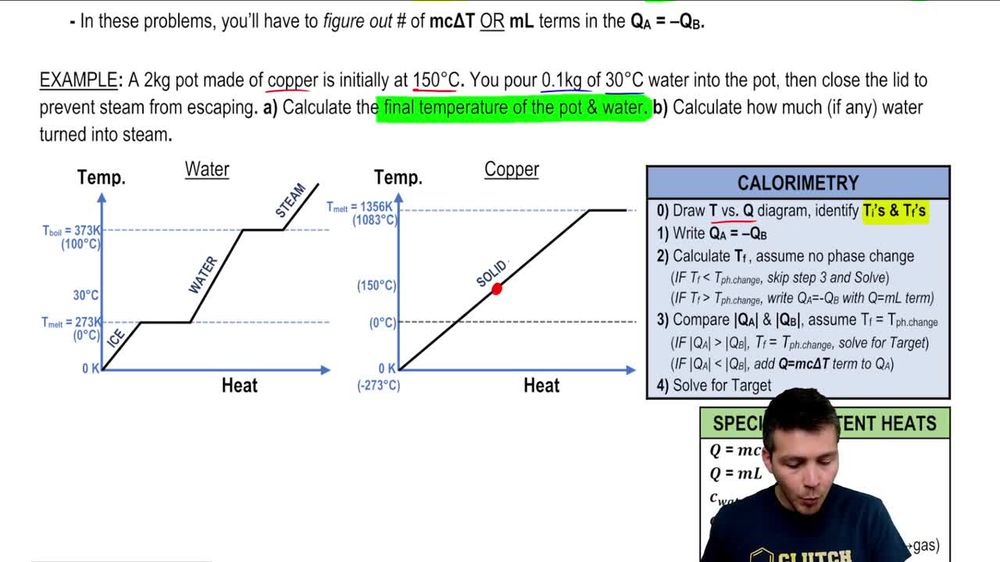
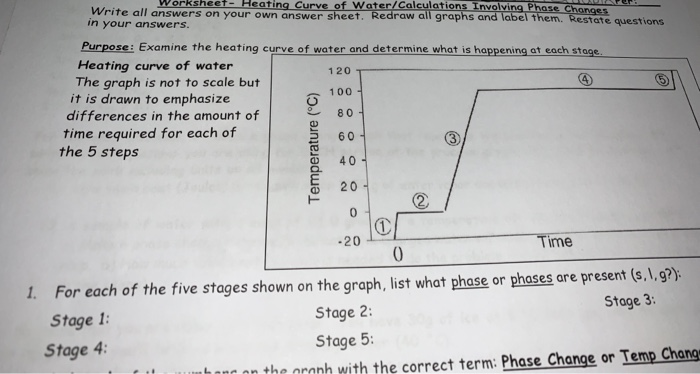
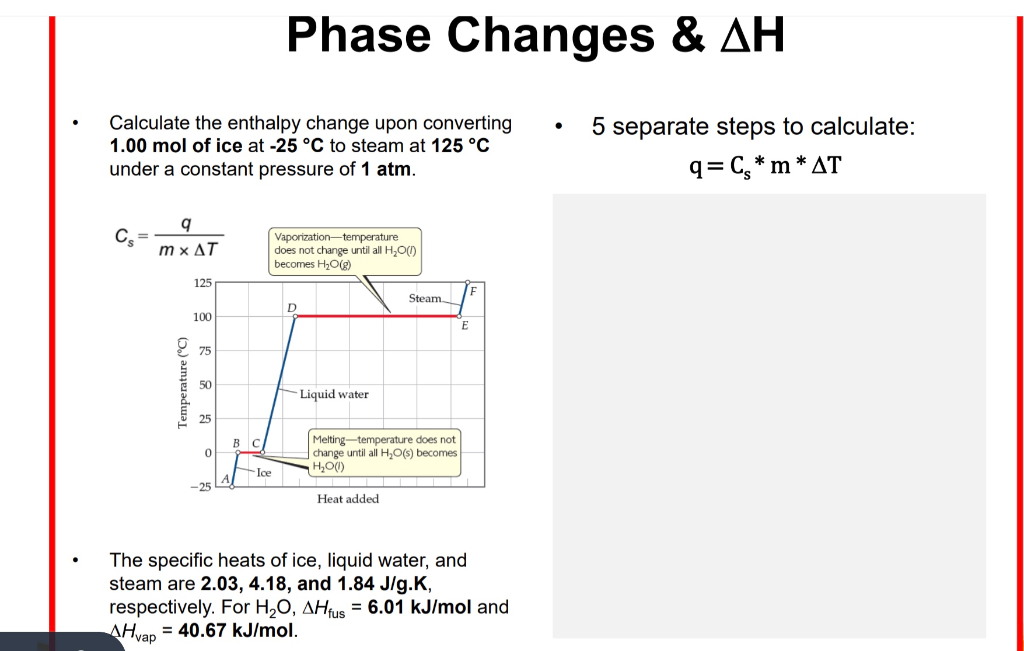
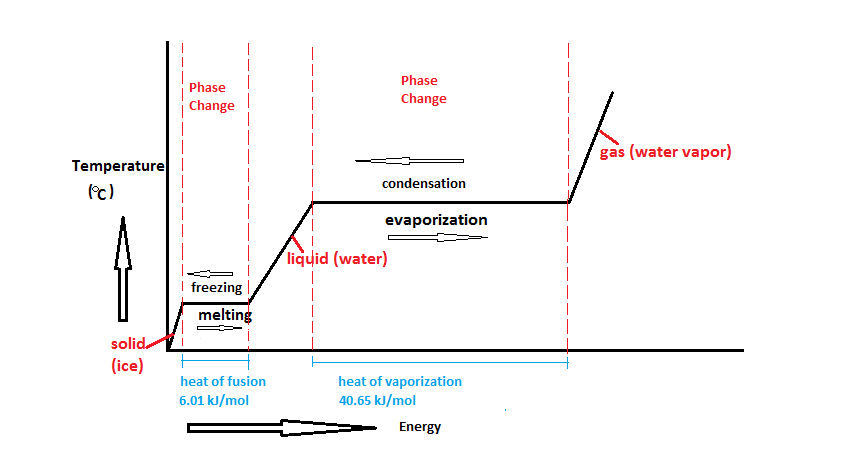
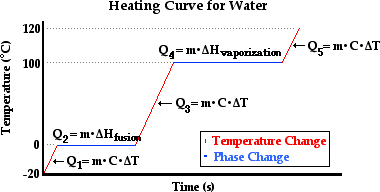
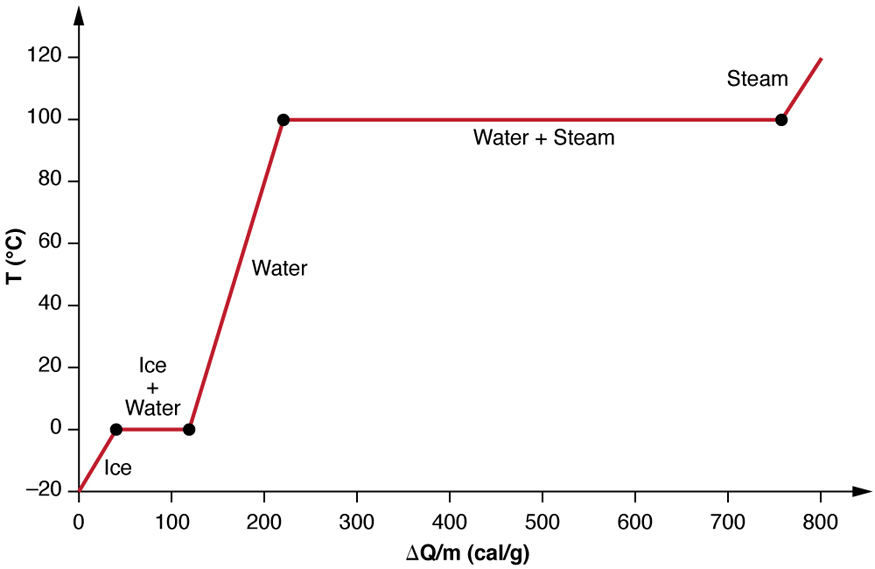
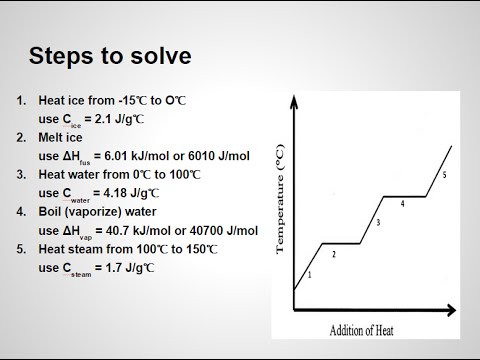
3.4 Solving Energy Problems Involving Phase Changes and Temperature Changes | METEO 300: Fundamentals of Atmospheric Science
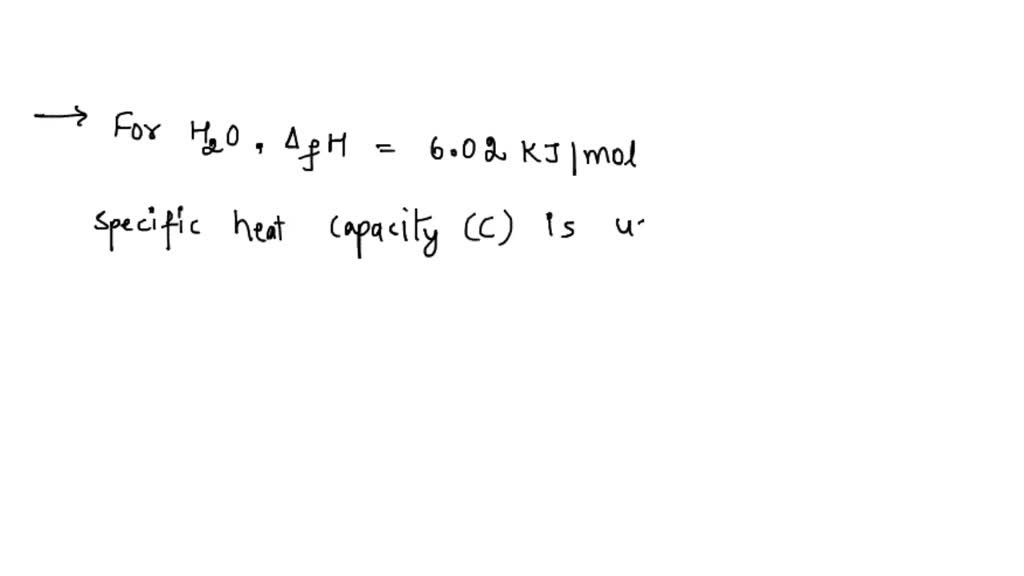
SOLVED: Heat of fusion (ΔHfus) is used for calculations involving a phase change between solid and liquid, with no temperature change. For H2O, ΔHfus=6.02 kJ/mol. Specific heat capacity (C) is used for