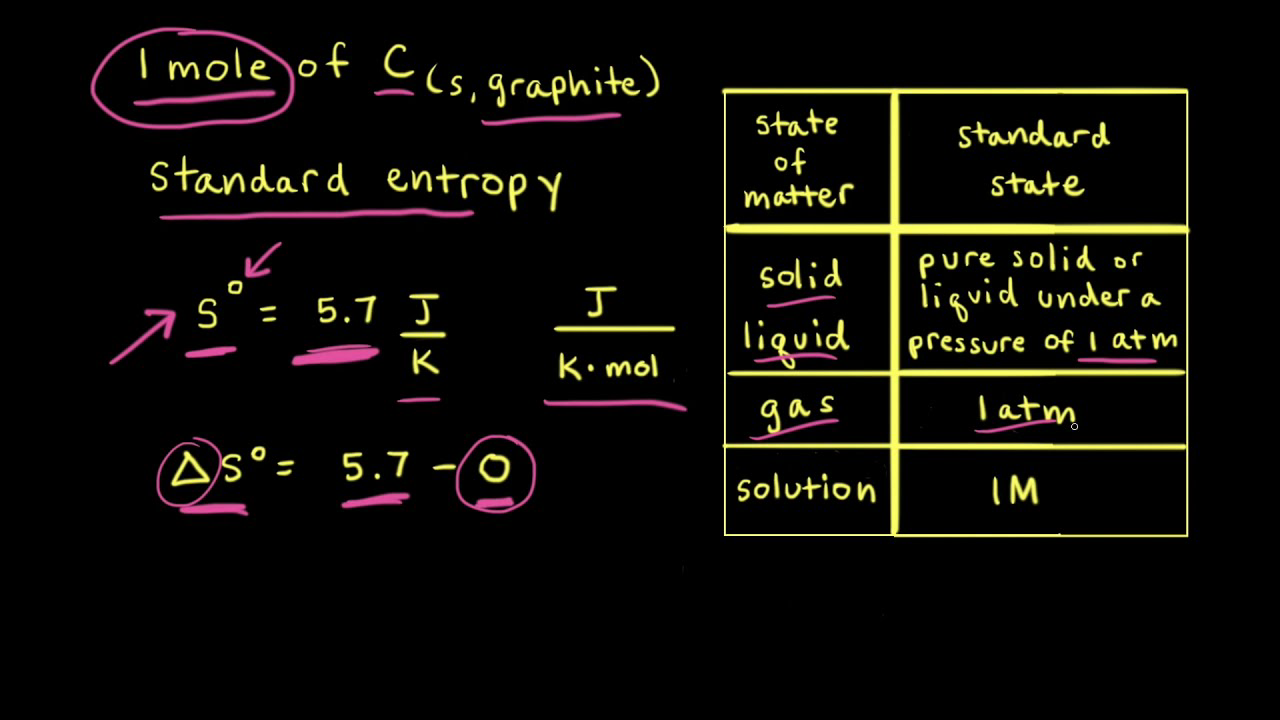
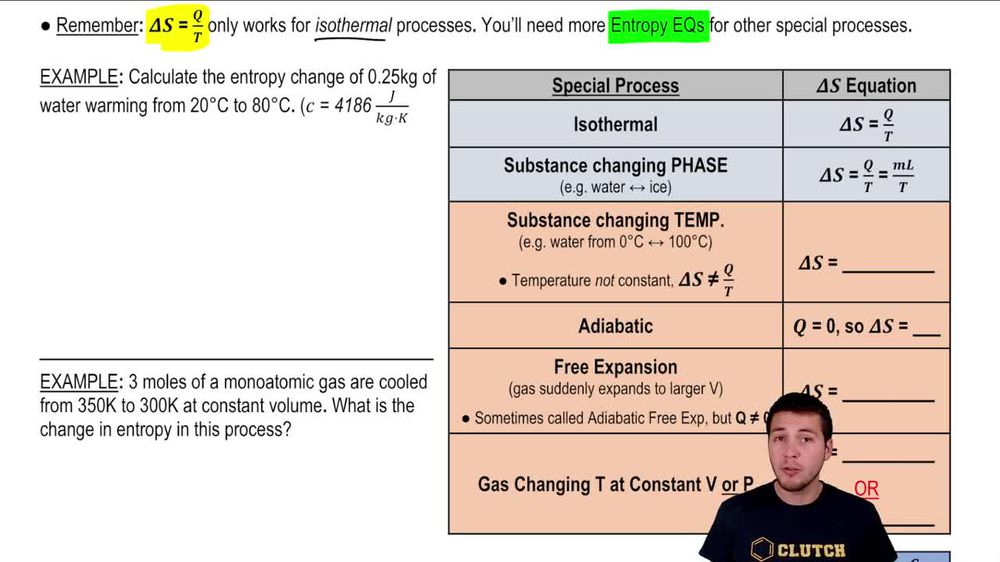
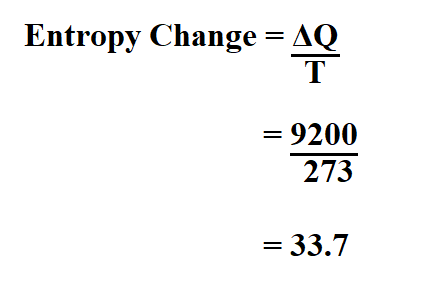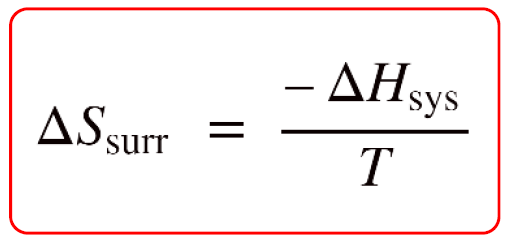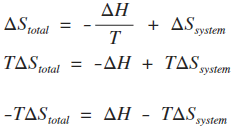Calculate the entropy change in surroundings when 1.00 mol of H2O (l) is formed under standard conditions at 298k, Given ,H^o = - 286kJ mol^-1

SOLVED: Calculate the entropy change of the universe (J/mol-K) when the entropy change of the system is 59.4 J/mol-K and the surroundings absorb 33.71 kJ of heat from the system at 77.74 °

Calculate the entropy change in surrounding when 1.00 mol of H2O(l) is formed under standard condition fH^ = - 286 KJ mol^-1 .
Calculate the entropy change in the system. and in the surroundings and the total entropy change in the universe when during - Sarthaks eConnect | Largest Online Education Community



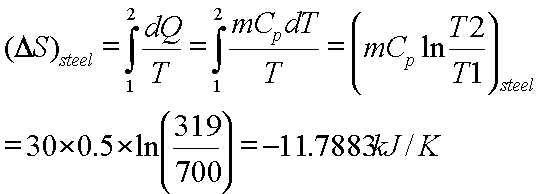


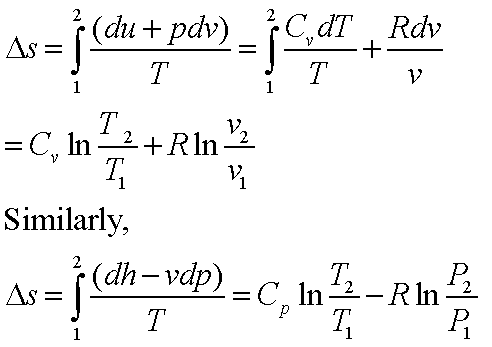



![15.2/R1.4 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube 15.2/R1.4 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/IwRy4iYVQLI/maxresdefault.jpg)