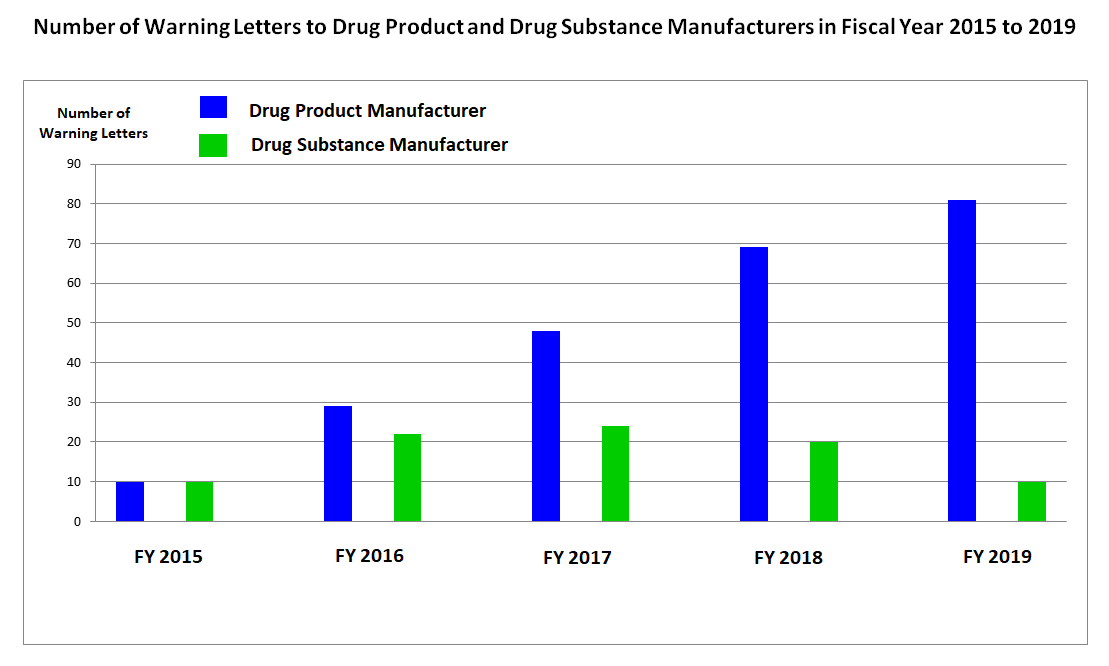
Batch Release without Determination of Identity and Strength and other GMP violations - A Look at FDA's Warning Letters over the Last Months - ECA Academy

Quality Transintegra on Twitter: "#WHO #GMP Training Basic GMP principles for Production areas https://t.co/35GmreHBML" / Twitter

Trans Integra on Twitter: ".#WHO #GMP Training slides. Basic requirements for personnel https://t.co/r0e4ViziD6" / Twitter

GMP Requests Removal of Cap on Powerwall and BYOD Home Battery Programs to Expand Customer Access to Cost-Effective Backup Power - Green Mountain Power

Standard Form of Agreement Between Owner and Construction Manager as Constructor where the basis of payment is the Cost of the Work Plus a Fee with a Guaranteed Maximum Price

Good Manufacturing Practice in China: Equipment Strategy and Quality Management to Compete with the West - BioProcess InternationalBioProcess International

Flammulina Velutipes Extract, Needle Mushroom Extract, GMP/HACCP Certificate - China Needle Mushroom Extract, Flammulina Velutipes Polysaccharide | Made-in-China.com




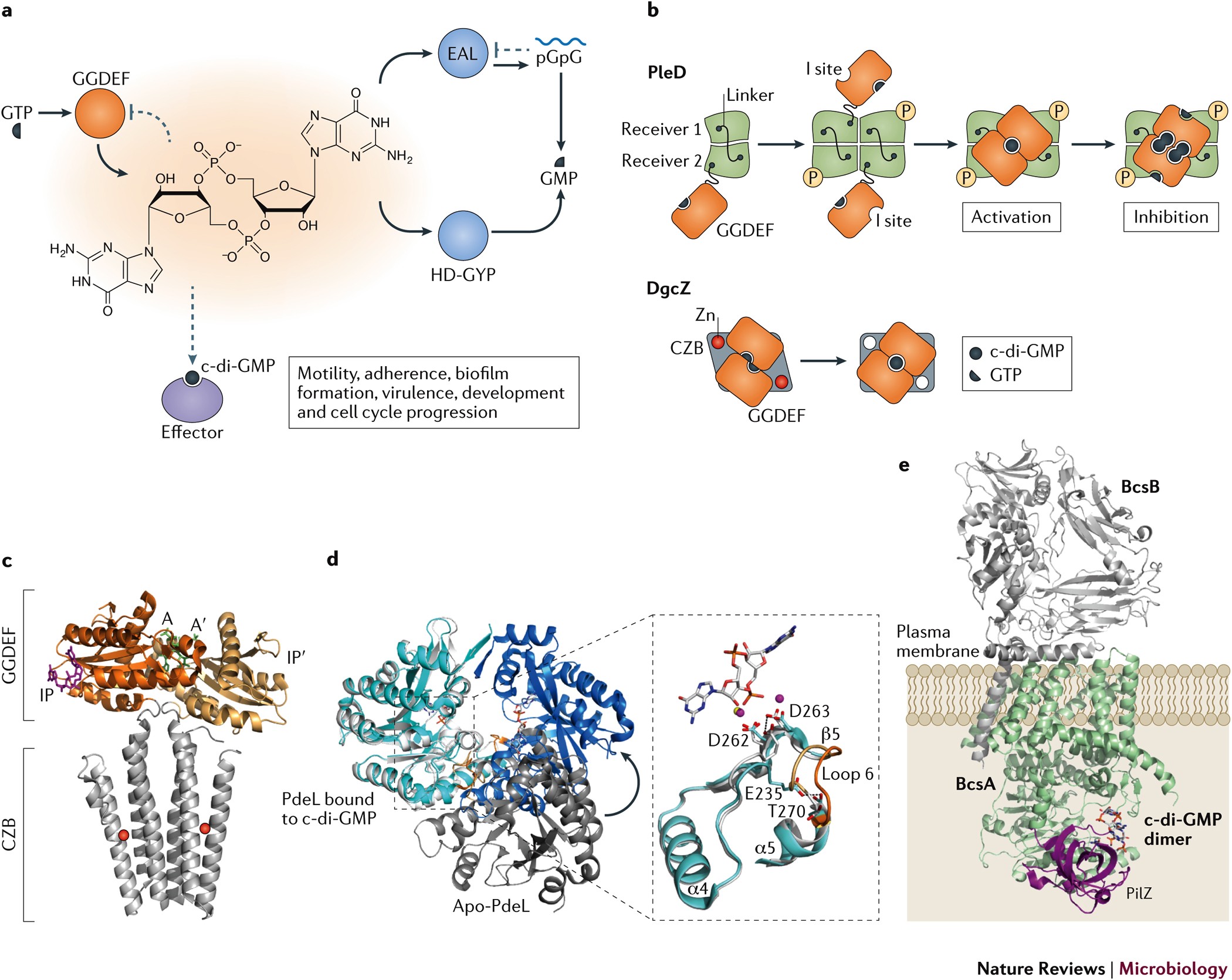





:quality(80)/f/54881/6720x4480/03237f2c2c/ar_2017_page23_08_barbara_hierweck1.jpg)